Preliminary Survey of Cave Fauna in the Phong Nha-Ke Bang World Heritage Site, Vietnam.
1GHD, Terrestrial and Marine Ecology, 239 Adelaide Tce Perth, WA 6000, Australia
2Head of Soil Ecology Department, Institute of Ecology and Biological Resources
Vietnam Academy of Science and Technology, 18 Hoang Quoc Viet RdCau Giay District, Hanoi, Vietnam
3PO Box 170, Yallingup, WA 6282, Australia
Email: 1 timothy.moulds@alumni.adelaide.edu.au 2 sacpd@iebr.ac.vn 3 reneemouritz@hotmail.com
Abstract
The Phong Nha-Ke Bang National Park (PNKBNP) is situated in the Quang Binh province of north central Vietnam. It was inscribed on the UNESCO World Heritage list in 2003 for its outstanding geomorphological features. The central park area is approximately 860 km2 and contains over 300 known caves, although to date no investigation of the fauna has occurred, despite the high likelihood of a significant endemic cavernicolous fauna.
The current preliminary survey was undertaken during May 2010 and provides an initial overview of the invertebrate fauna in two cave systems, Phong Nha Cave system (Phong Nha River Cave, Bi Ki Grotto, Fairy and Royal Grottos, and Tien Son Cave) and Dark Cave. These two caves are located approximately 5 km apart within a continuous block of karst. Due to the very limited amount of time available for the current preliminary survey it was decided to use active hand searching (hand foraging) to enable a wide variety of different habitats, and caves to surveyed quickly and detect the majority of species present within. The field component of the survey was conducted over five days, with multiple sites visited during each day.
The preliminary study of the cave fauna from the PNKBNP collected 248 individual specimens, representing at least 41 species from the three caves examined. The species include five (5) Classes, 14 Orders, and 29 families. Five species are common across the three caves, three spiders, a raphidophorid cricket and a millipede. The Dark Cave was found to contain many species that were not recorded from either Phong Nha or Tien Son caves, with 58% of species present found only from Dark Cave, compared with 55% in Phong Nha Cave and 44% in Tien Son Cave.
The survey collected 116 individual specimens from Phong Nha Cave comprising of four (4) classes, 10 orders, 19 families, and 20 species. The survey collected 41 individual specimens from Tien Son Cave comprising of four (4) classes, seven (7) orders, 12 families and 16 species. Diversity in the Dark Cave was high with five (5) classes, seven (7) orders, 15 families and 19 species recorded.
The presence of a blind scorpion in the wild section of Tien Son cave is highly significant, as there are currently only 20 described cave dwelling scorpions in the world that exhibit troglomorphic characteristics. This species is undoubtedly new to science and represents the first troglobitic scorpion in mainland Asia. This is the most significant discovery from the current preliminary survey of cave biodiversity in the PNKBNP.
The main differences in faunal assemblage shown by the current preliminary survey are the dramatic differences in faunal diversity and abundance between areas used by tourists and the wild sections of the same caves. The majority of fauna recorded from the Fairy and Royal Grottos are relatively common species found in several other areas. The abundance of the oniscoid isopod in these areas appears to be unnaturally high compared with Dark Cave populations, most likely a result of the rubbish and food scraps providing additional resources in this area.
The presence of bins within the caves also affects the faunal diversity, with virtually all the species recorded from the tourist section of Tien Son cave being located under or near bins. This fauna is likely to occur in much higher abundances than natural cave levels due to the additional food resources available. The lack of these food sources in the wild section of Tien Son cave resulted in much lower populations, or a complete absence, of species such as millipedes and staphylinid beetles, with only six (6) of the 16 species common between the two sections surveyed.
The results of the current preliminary study do not allow a meaningful comparison with other karst areas, either in Vietnam or the remainder of Asia. The preliminary results do, however, provide an indication of the diversity that may be revealed from this important karst area. The presence of undoubtedly the first cave dwelling troglomorphic scorpion from mainland Asia would suggest that the PNKBNP has a high potential for further important cave biology discoveries.
Introduction
The Phong Nha-Ke Bang National Park (PNKBNP) is situated in the Quang Binh province of north central Vietnam, inland from the city of Dong Hoi (Figure 1). It was inscribed on the UNESCO World Heritage list in 2003 for its outstanding geomorphological features. The central park area is approximately 860 km2 and contains over 300 known caves, including the largest currently known cave passage in the world Hang Son Doong Cave. The caves in the area were first explored and mapped in 1990 by a British expedition and this has continued to the present, with expeditions approximately every second year. This has resulted in 17 of the known caves in the Phong Nha area, and 3 in the Ke Bang area being mapped, (Figure 2) but to date no investigation of the fauna has occurred, despite the high likelihood of a significant endemic cavernicolous fauna.
Figure 1 Location of Dong Hoi within Vietnam (After www.vietnamtravel.com.au).
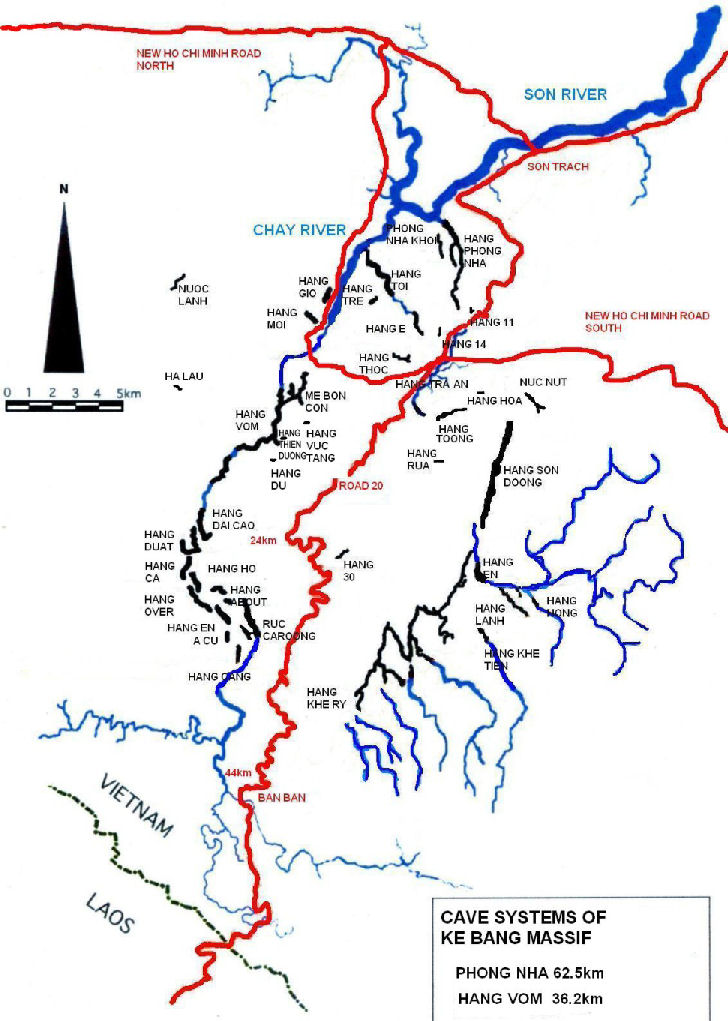
Figure 2 Map of Cave systems in the Ke Bang Massif (After www.vietnamcaves.com 2010).
Aims and Objectives of Preliminary Survey
The current preliminary survey aims to provide a basis for future biological surveys in the Phong Nha-Ke Bang World Heritage Site, Vietnam. The current preliminary survey aims to provide an initial overview of the invertebrate fauna in two cave systems, Phong Nha Cave system and Dark Cave. The Phong Nha Cave system for the purposes of this study consists of the Phong Nha River Cave, and includes three (3) tourist sections, Bi Ki Grotto, Fairy Grotto, and Royal Grotto. Tien Son Cave is situated approximately 200m above the entrance to Phong Nha Cave, and although no human sized connection is known, the caves are linked by micro and meso caverns which can be used by subterranean fauna . The Phong Nha and Dark Cave systems are located approximately 5 km apart within a continuous block of karst. The survey will aim to:
- Identify the majority of invertebrate fauna within each cave.
- The key habitats used by the fauna.
- Threats to the survival of the fauna.
- Provide management strategies which will ensure fauna survival.
- Compare and contrast the diversity of the two cave systems.
- Provide recommendations for future works to compliment the findings of the current study.
Introduction to Subterranean Biology
Caves form a very stable and generally homogenous environment in which to conduct various ecological and evolutionary experiments, such as on competition between species, resource partitioning, and the processes of speciation (Poulson and White, 1969). The total absence of light severely alters or completely removes many circadian cycles affecting ecosystem function (Lamprecht and Weber, 1992; Langecker, 2000). Temperatures are usually constant, varying only slightly between seasons. Humidity is commonly high, providing an ideal habitat for many invertebrate species susceptible to desiccation. The lack of photosynthetic plants changes the trophic structure of cave ecosystems, with energy sources usually being transported from the surface (Poulson and Lavoie, 2000; Poulson, 2005). Caves are defined as human-sized subterranean voids, although cave adapted animals are known to occur in the smaller spaces between large voids called micro- and meso caverns (Howarth, 2003).
Caves are divided into several distinct biological zones to aid interpretation (Figure 3). These correspond to the amount of available light and varying environmental conditions (Humphreys, 2000). The Entrance Zone is the area directly around the cave entrance; it is generally well lit, often supports photosynthetic plants, and undergoes daily temperature and humidity fluctuations. The Twilight Zone is just beyond the entrance zone and is often dominated by lichen and algae that require low light conditions. The temperature and humidity are still variable but fluctuations are dampened compared with epigean variation.
Deeper into a cave, light is reduced to zero and the Dark Zone is entered, which is subdivided into three zones, the transition, deep cave and stale air zones. The Transition Zone is perpetually dark, but still fluctuates in temperature and humidity determined by epigean conditions. The Deep Cave Zone is almost constant in temperature and humidity conditions. The Stale Air Zone is only found in certain caves and is an area of the deep zone that is constricted, and commonly contains elevated levels of CO2 and lower levels of O2 (Howarth and Stone, 1990).
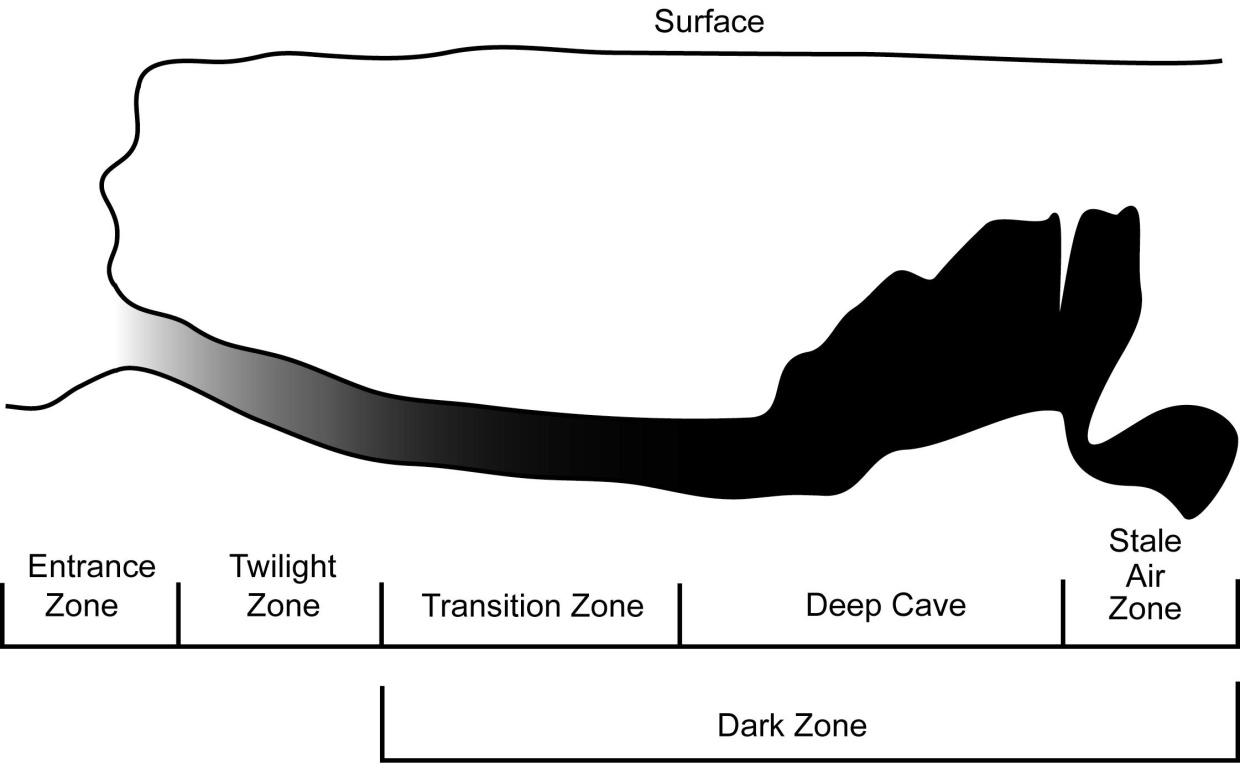
Figure 3 The environmental zones of a cave shown in cross section. After Moulds, 2006).
Classifications of cave dependence
Cave invertebrates are generally classified according to their degree of cave dependence using the Schiner - Racovitza system (Schiner, 1854; Racovitza, 1907), despite numerous other systems and variations being proposed and adopted by various authors (see references in Boutin, 2004).
The Schiner - Racovitza system classifies organisms according to their ecological association with subterranean environments, and relies upon detailed ecological knowledge of animals that is commonly lacking for most species. In order to circumvent this lack of knowledge, the concept of troglomorphy (Christiansen, 1962), specific morphological adaptations to the subterranean environment, is used to define obligate subterranean species. The term troglomorphy, initially confined to morphology has since been used to describe both morphological or behavioural adaptations (Howarth, 1973). This combination provides a practical system, easily applied in the field and with minimum of detailed ecological study required. The level of subterranean dependency for different ecological groupings is described below:
- Troglobites are obligate animals that possess specific adaptations (troglomorphisms) such as loss or reduction of pigmentation and/or eyes, flightlessness, elongate appendages and specific sensory adaptations (Barr, 1968; Poulson and White, 1969). These species rely solely on the cave environment for food and reproduction. They are generally restricted to the deep cave zone where conditions are the most stable and are rarely found closer to entrances in the twilight zone.
- Troglophiles are animals that can complete their entire lifecycle within a cave but possess no specific adaptations to the cave environment. These species are capable of living outside caves in suitably dark and moist epigean habitats.
- Trogloxenes are animals that regularly use caves for part of their lifecycle or for shelter, but must leave the cave to feed. Common examples of these are bats and cave swiftlets.
- Accidentals are animals that do not use caves on a regular basis and cannot survive in hypogean environments.
Aquatic hypogean animals are classified using a similar system to terrestrial hypogean animals except the prefix ‘stygo’ is used instead of ‘troglo’ (Humphreys, 2000).
The Trophic Basis of Cave Ecosystems
Cavernicolous populations are dependant for their survival upon energy inputs into cave systems. These inputs can vary widely, with availability of food usually being the primary limiting factor (Peck, 1976). Many cave ecosystems revolve around periodic flooding (Hawes, 1939; Humphreys, 1991; Culver et al., 1995) that carries organic material and accidental epigean animals into cave systems. Tree roots penetrating the roofs and walls are another energy source found commonly in tropical caves and lava tubes (Hoch, 1988; Hoch and Howarth, 1999). Guano from bats, birds and Orthoptera is an important energy source (Harris, 1970; Poulson, 1972; Decu, 1986; Blyth et al,. 2002; Moulds, 2004; Moulds, 2006) with large, varied and unique ecosystems existing around such deposits. Dead animals can be a source of food for scavengers near cave entrances (Richards, 1971). Accidentals wandering in from cave entrances also provide a food source, although this is generally periodic in nature and inconsistent in quantity, except in caves with large active rivers that are capable of carrying in large volumes of epigean animals, especially during high water flow periods.
For the most part, cave environments are generally depauperate in food and consequently are sparsely populated by cavernicolous animals. However, caves containing guano deposits differ fundamentally because there is a virtually unlimited food supply, commonly resulting in large populations of guano dependant arthropods known as guanobites. Guanobites possess no specific behavioural or morphological adaptations, presumably because of the lack of selection pressure to minimise energy expenditure that dominates the evolution of troglobites. The colonisation and establishment of guano dependent communities in caves is poorly understood. Mechanisms for the dispersal of guano dependent arthropods are potentially numerous, but most are poorly investigated at best (Moulds, 2004).
Sources and diversity of cave guano
Cave guano deposits from specific sources can each possess a unique assemblage of taxa (Horst, 1972; Poulson, 1972). Throughout the world’s biogeographic provinces different taxa are responsible for being the most important guano producers.
The most widespread and common guano is that produced by bats and these deposits are generally the largest in volume. The spatial and temporal deposition of bat guano differs from tropical to temperate caves. Cave-dwelling bats in temperate regions show an annual cycle of occupancy over summer months when pups are born, before colonies disperse to cooler, wintering caves where they enter torpor. This annual cycle results in large amounts of guano deposited over summer months and then a cessation of guano input for at least half the year. In contrast, tropical caves generally show constant bat occupancy rather than an annual cycle, and less aggregation of individuals due to warmer ambient temperatures (Trajano, 1996; Gnaspini and Trajano, 2000). Gnaspini and Trajano (2000) note that many bat populations in tropical Brazil are, however, commonly nomadic, resulting in roaming colonies varying their location in an irregular and non-seasonal fashion. This results in non-continuous guano deposition in a single locality over several years. The diet of bats (either haematophagous, insectivorous, frugivorous, or nectarivorous) also influences the composition of guano piles and, hence, the associated guanophilic communities (Gnaspini, 1992; Ferreira and Martins, 1998; Ferreira and Martins, 1999).
Birds are common guano producers in the northern parts of South America, the Caribbean and tropical caves of south-east Asia. Cave-dwelling birds nest in the dark zone, providing an important energy resource for many cavernicolous animals. Swiftlets (Aerodramus spp.) nest in the entrance and dark zones of tropical caves in south-east Asia, northern Australia and the Pacific, and are insectivorous (Medway, 1962; Humphreys and Eberhard, 2001; Koon and Cranbrook, 2002). The volumes of bird guano deposited are comparable to similar sized bat populations.
Survey Timing and Participants
The survey was conducted between the 13th – 23rd May 2010. The survey was undertaken by a specialist cave biologist, Dr Timothy Moulds (Australia), and assisted by a Vietnamese colleague Dr Pham Dinh Sac. Additional assistance was provided by Renee Mouritz (Australia) who has experience in cave interpretation, guiding and speleology. Further field assistance was provided by Nguyen Ngoc (Phong Nha - Ke Bang National Park Tourism Centre), Dang Ngoc Kien (Scientific Research and Wildlife Rescue Centre), Dr Joachim Esser (GTZ), Nguyen Thi Phuong Ha (GTZ), and Dirk Euller (German Development Service, DED).
Report Limitations and Exclusions
The current report was produced during a 10 day visit to the PNKBNP in May 2010. The survey was intended as a preliminary investigation into the subterranean biodiversity of two of the cave systems within the park. Identification of specimens collected were undertaken with limited reference material and are considered to be preliminary identification for the purposes of the report. This study was limited to the requirements specified by the client and the extent of information made available to the consultant at the time of undertaking the work. Information not made available to this study, or which subsequently becomes available may alter the conclusions made herein. The survey was planned to include an assessment of the Paradise Cave, but due to construction work currently being undertaken at the cave entrance, access was unavailable.
Survey Methodology
Surveys for subterranean fauna may use many different techniques according to the type of fauna being targeted and the amount of time available for the survey. These methods can include:
- pitfall traps (baited and unbaited).
- hand foraging (using forceps and paintbrushes to actively collect observed fauna).
- litter traps left in situ for days or weeks and then fauna extracted in a tullgren funnel.
- net hauling of water for aquatic fauna.
- nets left in situ in narrow streams to sieve water flows for discrete time periods.
Due to the very limited amount of time available for the current preliminary survey it was decided to use active hand searching (hand foraging) to enable a wide variety of different habitats, and caves to be surveyed quickly and detect the majority of species present within. In order to undertake a more comprehensive survey of the subterranean fauna (vertebrate and invertebrate) a combination of multiple techniques in each cave over longer time periods would be required. This was beyond the scope of the current project.
The field component of the survey was conducted over five days in May 2010, with multiple sites visited during each day. The sites visited are listed below:
- Phong Nha Cave – Fairy and Royal Grottos
- Phong Nha Cave – Fairy Cave Swiftlet guano deposit
- Phong Nha Cave – Bi Ki Grotto
- Phong Nha Cave – River Banks
- Phong Nha Cave – Rockpile at end of 1st river section
- Tien Son Cave – Tourist section
- Tien Son Cave – Wild section
- Dark Cave – Twilight Zone
- Dark Cave – Transition Zone
Material collected was placed in 100% ethanol for preservation, and sorted using an Olympus SZ-60 stereomicroscope. Specimens were identified to lowest practical taxonomic level using the resources available at the time of the survey in Dong Hoi. Araneae were identified by Dr Pham Dinh Sac and all remaining material was identified by Dr Timothy Moulds. All material collected remains the property of the Peoples Republic of Vietnam, and has been handed over with a signed protocol to the Scientific Research and Wildlife Rescue Centre of PNKBNP.

Figure 4 Renee Mouritz and Nguyen Ngoc searching for cave invertebrates, Tien Son Cave. (Photo Tim Moulds).
Survey Results
The preliminary study of the cave fauna from the PNKBNP collected 248 individual specimens, representing at least 41 species from the three caves examined. The species include five (5) Classes, 14 Orders, and 29 families. Five species are common across the three caves, three spiders, a raphidophorid cricket and a millipede. The Dark Cave was found to contain many species that were not recorded from either Phong Nha or Tien Son caves, with 58% of species present found only from Dark Cave, compared with 55% in Phong Nha Cave and 44% in Tien Son Cave.
The rhaphidophorid cave crickets, although not formally recorded from every cave or site were present at every location, however, only adult specimens were collected when present.
The results are presented individually for each cave surveyed, Phong Nha, Tien Son, and Dark Cave. A detailed table of results is presented in Table 1.
|
|
|
|
|
|
Phong Nha Cave |
|
|
|
|
Tien Son Cave |
|
Dark Cave |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Class |
Order |
Family |
Subfamily / Genus |
Species |
Fairy/Royal Grotto |
Fairy Grotto Guano |
Bi Ki Grotto |
River Edge |
Rockpile |
Tourist Section |
Wild Section |
Twilight Zone |
Transition Zone |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Oligochaeta |
|
|
|
sp. 1 |
|
|
|
|
|
1 |
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
sp. 2 |
|
|
|
|
|
|
|
1 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Crustacea |
Isopoda |
Oniscoidea? |
|
sp. 1 |
15 |
|
|
|
|
|
|
6 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Philoscoidea? |
|
sp. 1 |
|
|
|
|
|
|
|
4 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Decapoda |
|
|
sp. 1 |
|
|
1 |
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Arachnida |
Araneae |
Amaurobiidae |
Platocoelotes |
sp. 1 |
|
|
|
|
|
|
|
|
4 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Araneidae |
Araneus |
sp. 1 |
5 |
|
3 |
5 |
11 |
2 |
2 |
2 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Araneus |
sp. 2 |
|
|
|
|
|
|
4 |
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Araneus |
sp. 3 |
|
|
|
|
|
|
|
|
6 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Ctenidae |
|
sp. 1 |
|
|
|
|
1 |
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Gnaphosidae |
Gnaphosa |
sp. 1 |
1 |
2 |
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Linyphiidae |
Neoscona |
sp. 1 |
1 |
1 |
3 |
9 |
1 |
3 |
2 |
1 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Neoscona |
sp. 2 |
|
|
|
|
|
|
|
|
12 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Lycosidae |
Lycosa |
sp. 1 |
2 |
|
|
|
|
1 |
|
1 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Ochyroceratidae |
Speocere |
sp. 1 |
|
|
|
|
1 |
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Sparassidae |
Heteropoda |
sp. 1 |
4 |
1 |
1 |
3 |
2 |
1 |
3 |
|
3 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Sinopoda |
sp. 1 |
|
|
|
|
|
|
3 |
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Sinopoda |
sp. 1 |
|
|
|
|
|
1 |
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Symphytognathidae |
Patu |
sp. 1 |
|
|
|
|
5 |
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Patu |
sp. 2 |
|
|
|
|
|
|
3 |
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Telemidae |
Telema |
sp. 1 |
|
|
|
|
|
|
|
|
6 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Telema |
sp. 2 |
|
|
|
|
|
|
|
|
2 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Tetrablemmidae |
Lehtinenia |
sp. 1 |
|
|
|
|
|
|
2 |
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Theridiidae |
Theridiion |
sp. 1 |
|
|
|
|
2 |
|
|
|
7 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Scorpionida |
|
|
sp. nov. |
|
|
|
|
|
|
2 |
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Opilione |
|
|
sp. nov. |
|
|
|
|
|
|
|
4 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Myriapoda |
Julida? |
|
|
sp. 1 |
|
|
|
|
|
1 |
1 |
|
1 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Diplopoda |
|
|
sp.1 |
|
|
|
15 |
2 |
9 |
13 |
2 |
1 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Diplopoda |
|
|
sp.2 |
|
|
|
|
|
|
|
|
1 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Insecta |
Collembola |
Entomobryidae |
|
sp. 1 |
|
|
|
1 |
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Orthoptera |
Rhaphidophoridae |
|
sp. nov. |
4 |
|
1 |
1 |
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Coleoptera |
Staphylinidae |
Aleocharinae |
sp. 1 |
|
2 |
|
|
|
1 |
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
Oxytelinae? |
sp. 1 |
|
|
|
|
|
2 |
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Coleoptera |
|
Staphylininae |
sp. 2 |
|
|
|
|
|
|
|
|
1 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Ptiliidae |
|
sp. 1 |
|
|
|
|
|
6 |
1 |
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Lepidoptera |
Tineidae? |
|
sp. 1 |
|
1 |
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Hymenoptera |
Ichneumonidae |
|
sp. 1 |
|
1 |
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Chalcidoidea |
|
sp .1 |
|
4 |
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Diptera |
Anthomyiidae |
|
sp. 1 |
|
1 |
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. Species diversity and abundance from three caves in the Phong Nha – Ke Bang National Park, Quang Binh Province, Vietnam.
Phong Nha Cave
The survey collected 116 individual specimens from Phong Nha Cave comprising of four (4) classes, 10 orders, 19 families, and 20 species.
Fairy and Royal Grottos
This area of Phong Nha Cave contained seven (7) species, of which five (5) are spiders. The remaining species are Oniscoid isopods (slaters), and the ubiquitous cave crickets. The abundance of slaters was greater near the beach sections of Fairy Cave where ground disturbance is lower and also near rubbish bins in the remainder of the cave. Cave crickets were also more abundant near the beach area and away from main foot traffic areas.
The Fairy Cave also contains a deposit of swiftlet guano on an elevated portion of wall, that does not get trampled by foot traffic. This small (1m x 0.4m) deposit contains considerable diversity compared with the remainder of the cave, and was found to contain 3 species of spider which are also found elsewhere in the Fairy Grotto, but more importantly an additional six (6) species not found anywhere else in the Phong Nha Cave. The diversity of the small guano deposit is very different to that observed in any other cave habitat examined during the survey, and included three (3) orders and four (4) species found nowhere else during the survey of the three (3) caves examined.

Figure 5 Swiftlet guano deposit in the Fairy Grotto contained considerable diversity not found in other cave habitats. (Photo Tim Moulds).
Bi Ki Grotto
This section of the Phong Nha cave contained the lowest diversity of all the areas examined for cave fauna, with 5 species, and also the lowest abundance of specimens collected, with just 9 individuals recorded. All the species recorded here were also found elsewhere, apart from the freshwater crab, which is considered a surface (epigean) species that is only able to survive here due to the constant light available.
Phong Nha River Passage
Several sand banks on the side of the main Phong Nha River passage were examined during the study and considerable diversity was recorded from these sites. Six (6) species were recorded from this habitat, half of which are spiders found in numerous different cave habitat areas examined during the study. One taxa, an Entomobryid Collembola was found in the river sand beaches and no other areas in the Phong Nha cave, although a longer and more detailed survey may reveal their presence to be more widespread.
Rockpile Section
The end of the first river section of Phong Nha cave, at approximately 2km from the entrance contains a large rockpile chamber. This area of the Phong Nha Cave was found to contain eight (8) taxa, of which seven (7) are spiders, and the other species recorded was the relatively common millipede with reduced eyes. No other insects or crustaceans were recorded from this locality.
Tien Son Cave
The survey collected 41 individual specimens from Tien Son Cave comprising of four (4) classes, seven (7) orders, 12 families and 16 species.
Tien Son Cave - Tourist Section
The tourist section of Tien Son cave encompasses the entrance, twilight and beginning of the transition zone of the cave (refer Section 1.3). This area did contain considerable diversity, with 11 taxa recorded, however, it must be noted that the vast majority of this was located under or in the vicinity of rubbish bins near the end of the tourist section. Much of the abundance of both millipedes and beetles was also recorded from these areas. Spider diversity was also mostly restricted to isolated areas away from main walking paths and foot traffic areas, where extensive trampling of floors has severely impacted upon available habitat for invertebrates.

Figure 6 Sparassidae Heteropoda sp. 1 from Tien Son Cave, leg span approximately 15 cm (Photo Tim Moulds).
Tien Son – Wild Section
The wild section of Tien Son Cave contains noticeably different habitat to the tourist section due to far less trampling of floor habitats, and it correspondingly contains a different fauna assemblage. This habitat contains the same species richness as the tourist section (11 species), but five (5) of the species recorded here were not found in any other cave sections surveyed. This includes four (4) spiders and a blind scorpion.
The presence of a blind scorpion in the wild section of Tien Son cave is highly significant, as there are currently only 20 described cave dwelling scorpions in the world that exhibit troglomorphic characteristics (Volschenk and Prendini, 2008). This species is undoubtedly new to science and represents the first troglobitic scorpion in mainland Asia. This is the most significant discovery from the current preliminary survey of cave biodiversity in the PNKBNP.

Figure 7 Blind scorpion from Tien Son Cave, Body length approximately 10 mm. (Photo Tim Moulds).
Dark Cave (Hang Toi)
The Dark Cave is situated approximately five kilometres to the west of the entrance of Phong Nha cave on the banks of the Chay River. The cave is a large streamway outflow cave, with minor side passages. The current survey recorded 19 species from the Dark Cave, comparable to both Phong Nha (20 species) and Tien Son Caves (16 species), however, it should be noted the relative survey effort in Dark Cave was considerably lower than that expended in the Phong Nha system.
Diversity in the Dark Cave was high with five (5) classes, seven (7) orders, 15 families and 19 species recorded.
Twilight Zone
The twilight zone of the cave contained some species common across the caves examined including rhaphidophorid crickets, millipedes and what appeared to be the same morpho-species of oniscoid isopod as seen in Fairy Grotto, however, in much lower abundances than in Phong Nha. This species was associated with a small deposit of bird guano, but no other guano associated species were found here, possibly due to the high energy seasonal flooding that occurs here, stopping the establishment of a large and diverse guano community. This locality was also the only site where opiliones were collected.

Figure 8 Oniscoid slater in small bird guano deposit in the twilight zone of Dark Cave. (Photo Renee Mouritz).
Transition Zone
The transition zone of Dark Cave consisted of a small side passage which was subject to seasonal flooding. This passage contained considerable amounts of organic material which attracts a diverse range of 1st order consumers including passalid beetles, millipedes, and crickets. This resulted in a very diverse spider assemblage with seven (7) species recorded, and five (5) of these not found in any other caves examined during the current study.
Cave Biodiversity Discussion
Comparison of Cave Biodiversity in Phong Nha-Ke Bang NP
The three caves examined show similarity in diversity, as well as some distinct differences, based upon the hydrology, morphology and age of the caves. The Phong Nha Cave System (Royal and Fairy Grottos, Bi Ki Grotto, and the main Phong Nha river cave passage) and Tien Son Cave, situated approximately 200m above the current river level of Phong Nha Cave, do not have a human sized connection, however, cave fauna can invariably move between the caves via the network of meso and micro caverns that are present in all karstic systems.
The main differences in faunal assemblage shown by the current preliminary survey are the dramatic differences in faunal diversity and abundance between areas used by tourists and the wild sections of the same caves. Phong Nha cave contains three tourist sections, Bi Ki Grotto, and the contiguous Royal and Fairy Grottos near the cave entrance. These areas are extremely heavily impacted by foot traffic over every horizontal surface, with no defined paths through much of the tourist route. This has resulted in limited habitat remaining that is capable of supporting cave fauna. The large amount of rubbish in the tourist sections, and the presence of rubbish bins within the caves have also affected the assemblage of cave fauna in these areas. The majority of fauna recorded from the Fairy and Royal Grottos are relatively common species found in several other areas. The abundance of the oniscoid isopod in these areas appears to be unnaturally high compared with Dark Cave populations, most likely a result of the rubbish and food scraps providing additional resources in this area.
The presence of bins within the caves also affects the faunal diversity, with virtually all the species recorded from the tourist section of Tien Son cave being located under or near bins. This fauna is likely to occur in much higher abundances than natural cave levels due to the additional food resources available. The lack of these food sources in the wild section of Tien Son cave resulted in much lower populations, or a complete absence, of species such as millipedes and staphylinid beetles, with only six (6) of the 16 species common between the two sections surveyed.
The cave fauna recorded from the Dark Cave showed the greatest difference to both Phong Nha and Tien Son caves, due to both the different cave morphology and the absence of tourism within the Dark Cave. While the overall species diversity was not significantly greater, the species recorded from Dark Cave were mostly not recorded from either of the other caves surveyed. It is impossible to confirm the cause of this difference with the current preliminary data, however, it is likely to be a combination of both natural faunal compositions and absence of impacts to the available habitat within Dark Cave compared with Phong Nha and Tien Son Caves.
Regional Significance
The results of the current preliminary study do not allow a meaningful comparison with other karst areas, either in Vietnam or the remainder of Asia. As the specimens collected are identified further and additional, more comprehensive surveys are carried out in the PNKBNP the true significance of the areas subterranean biodiversity will become apparent. The preliminary results do, however, provide an indication of the diversity that may be revealed from this important karst area. The presence of undoubtedly the first cave dwelling troglomorphic scorpion from mainland Asia would suggest that the PNKBNP has a high potential for further important cave biology discoveries. The sheer amount and diversity of caves within the area provide extensive and varied habitats to support a wide variety of different subterranean communities, that when properly studied will likely reveal a rich and important diversity for Vietnam and the PNKBNP World Heritage Site.
Recommendations to Preserve Cave Biodiversity
Cave Management
People going off the designated path have the potential to damage cave habitat. People leaving rubbish in the cave, spitting, toileting in the cave all create artificial food sources which attract outside fauna. Noise levels in the cave are also disturbing the bat and swiftlet populations which reside in the cave, relying upon the cave for shelter.
Noise/Uncontrolled Behaviour
Both bats and swiftlets use Phong Nha Cave and Tien Son Cave as a roosting/nesting ground. This was determined through visual observation of the animals and the presence of their droppings or guano. Old guano deposits found in areas of the caves indicate that bat and swiftlet population numbers were much higher in the past than those currently. The excessive noise in the cave, due to the excited nature of people, are having an impact on the natural assemblage of bat and swiftlets. These animals both provide a very important source of energy for many invertebrate species within the cave system. A change in bat and swiftlet populations will result in a dramatic alteration to the faunal assemblage seen in the caves. The following actions will aid in reducing noise:
- Staff training – all staff must be made aware of the importance of maintaining low noise levels inside the caves.
- Ensure staff have the authority to maintain low noise levels in the caves.
- Adequate signage to educate visitors about the bat and swiftlet populations and the importance of their presence to a healthy cave and surface ecosystem (they eat insects outside the caves). and the need for them to keep quiet so as not to further disturb them.
Rubbish (Accumulation of rubbish within the caves)
There is little to no enforcement of the park rules about eating, drinking, and smoking in the caves. This is resulting in large amounts of rubbish being found throughout the cave including plastic drink bottles, fruit juice cartons, beer and soft drink cans, good luck money, footwear, clothing, fruit peel, eggshells, and peanut shells. These artificial food sources have the potential to attract pest species into the caves, artificially altering community structures and greatly impacting on the natural species diversity in the cave systems.
The cave survey found evidence of rat presence in Tien Son Cave. It is obvious that eating and drinking in the caves is a huge problem and needs to be stopped. Rubbish bins are in place throughout Tien Son Cave and Phong Nha Cave and are to a point being used but they in themselves act as an artificial food source as the faunal assemblage found around the base of one showed. It is highly recommended that the rubbish bins are removed from the cave, an extensive cave clean up is done to remove rubbish that has been disposed of inappropriately throughout the caves and the rules of not eating and drinking within the cave are strictly enforced, as that of no smoking.
- Staff training – all staff must be made aware of why the worldwide accepted rules of No Smoking, No Eating and No Drinking within the cave are so important and be given guidance in how to enforce them.
- Conduct a thorough clean up of the caves with ALL rubbish removed and disposed off appropriately.
- Ensure staff have the authority to prohibit visitors from carrying food and drink items into the caves .
- Remove rubbish bins as they are acting as artificial food sources and should no longer be necessary if people are not eating and drinking in the caves.
- Instead of ‘good luck’ money make it good luck to pick rubbish up from the cave and take it outside to the bin.

Figure 9 Peanut shells discarded in Tien Son Cave creates artificial food sources that attract surface species and enable rats to live inside the cave. (Photo Tim Moulds).

Figure 10 Cigarette butts and other rubbish in Bi Ki Grotto. Rubbish provides food for pest species such as rats inside the cave. (Photo Tim Moulds).
Inappropriate Lighting
The current lighting system in both Phong Nha and Tien Son is not conducive to creating a suitable habitat for cave fauna. The constant light is having a detrimental effect on the bat and swiftlet populations in the caves. The current lighting system also creates a fantasy world cave experience that is a potential source of the excitement (exhibited as noise) felt by visitors in the cave. This noise will be of great disturbance to the bat and swiftlet populations within the cave systems.
Lampenflora is also another problem resulting from the inappropriate lighting in the caves. The growth of algae, moss and in extreme cases ferns deep into the cave system again has the potential to create an artificial food source.
These problems can be greatly improved by reducing the amount of light being used as well as the type of light used and duration of time that the lights are left on throughout the cave (as per recommendations made by Brian Clark, 2009a).

Figure 11 Lampenflora growth on a speleothem inside Tien Son Cave. This not only damages the speleothem but also creates an artificial food source in the cave that attracts surface species into the cave. (Photo Tim Moulds).
Floor – habitat destruction
Due to the undefined nature of the pathways in both Tien Son Cave and the Phong Nha Cave the floor of the cave has been badly trampled with any suitable habitat destroyed as a result. It is imperative that the movement of visitors through the caves be restricted to distinct pathways. Ideally these would be elevated to prevent further damage to the cave floor but in the early stages roping off walkways would be an effective and cheaper option.
- Staff training and control/enforcement of where visitors walk.
- Roping off walkways to define where tourists are allowed to walk.
- Ultimately developing a series of elevated pathways to further minimise any impact on the cave floor and to allow habitat to reinstate itself.
- Clean the cave of both rubbish and compacted mud on floor areas to further reinstate habitat for cave fauna.

Figure 12 Floor compaction in Phong Nha Cave. (Photo Tim Moulds)

Figure 13 Floor compaction in Tien Son Cave. (Photo Tim Moulds).
Summary of Key Management Recommendations to Preserve Cave Biodiversity
The preliminary survey results lead to several very important recommendations to ensure the caves maintain their biodiversity in the future:
- Immediately make defined pathways in the tourist caves marked by posts and rope to stop the cave floors being destroyed by people walking on them. This destroys habitat for many insects and spiders that live in the cave. Ultimately raised walkways should be installed in all tourist areas to minimise impact to cave floors.
- Clean the compacted mud from the cave floor to allow habitat for cave insects and spiders to recover.
- Clean up the rubbish left throughout the cave. Rubbish attracts rats into the cave that will eat cave insects and spiders.
- Remove the rubbish bins which attract rats (see above), and also attracts surface insects which are affecting the diversity and abundance of cave insects and spiders.
- Stop people eating and drinking inside the caves as food scraps dropped encourage rats to live in the caves.
- Reduce noise in the caves as this is disturbing the bat and swiftlet populations which support diverse insect communities in the caves. If the noise continues, the bats and swiftlets may leave the cave permanently, destroying the cave guano insect ecosystem.
- Enforce the ‘No Smoking’ ban inside the caves as this is also disturbing the bat and swiftlet populations, and also increases rubbish dropped on floors as butts and empty packets.
- Change the lighting in the caves as it is causing the growth of lampenflora (plants that grow under artificial light in caves). The lampenflora is providing an artificial food source for surface species not usually found in caves, and thus affecting the diversity and abundance of cave insects and spiders.
- Additional, more comprehensive biodiversity surveys are required in order to fully appreciate the diversity of cave fauna in the PNKBNP. This will provide a far greater understanding of biodiversity and the unique species present here.
- Prior to the development of additional caves for tourism, they must be comprehensively assessed for cave biodiversity to provide baseline data to enable annual monitoring of impacts to fauna. The baseline survey will also identify any key habitats to be preserved within each cave, and any important species that may require special protection.
These recommendations (1 - 8), are the same as those made by Brian Clark (Clark, 2009a, 2009b), to stop the destruction of the caves and formations and ensure World Heritage Values are maintained, but are made here in order to ensure the biodiversity values of the caves are preserved.
Recommendations for Future Work
The current study provides only a very preliminary study of three easily accessible caves within the large PNKBNP. The diversity contained in these caves is invariably much greater and considerably more survey effort will be required before a fuller understanding of the caves true biodiversity values can be ascertained. With this in mind a series of future studies are recommended below to build upon the information gathered during the current survey.
Key recommendations for future cave biodiversity studies are:
- The specimens collected during the current survey should be held by an appropriate research institute with suitable laboratory space and access to specialised library resources such as the Hanoi University to enable their continued identification and study by taxonomic experts. Due to the limitations in both time and available local resources in Dong Hoi the level of identification of the material collected during the current survey is preliminary and considerable further work is required to determine the number of species new to science collected. This collection can then form the basis for any future surveys to be conducted on the cave fauna of PNKBNP.
- Undertake a literature review of cave fauna known from PNKBNP including any references in cave exploration reports of the area. This could also include the whole of Vietnam, due to the general absence of records making this a readily achievable task. This would enable the biodiversity in PNKBNP to be placed and ranked in a local and regional context, and provide a ‘state of play’ for additional work to be undertaken effectively.
- A follow up survey of the Phong Nha Cave system (Phong Nha and Tien Son Caves) 12 months after any cave cleaning and installation of walkways have been undertaken. This will enable an assessment of the cave biodiversity and effects of rehabilitation. Monitoring of cave fauna should be repeated annually to provide an ongoing assessment of the effectiveness of the rehabilitation actions including; installation of defined walkways, rubbish removal, alteration of lighting and cleaning of lampenflora, smoking ban enforced, etc. This should be undertaken by a specialist in cave biology in conjunction with a local Vietnamese organisation with experience in cave biodiversity, ideally with previous experience in the PNKBNP karst.
- Undertake a comprehensive biological survey of three to four caves in the PNKBNP situated in different hydrogeologic areas to provide a greater understanding of cave biodiversity within the entire park. This should include caves from both the main drainages identified in the Park.
Conclusions
The present study has undertaken a preliminary investigation of three easily accessible caves situated on the edge of the karst contained within the Phong Nha – Ke Bang National Park World Heritage Site. This small study will provide a basis for more detailed work on the diversity of the PNKB karst in the future by providing an indication of the potential for endemic fauna and rare and significant species to be identified. In order to properly understand the biodiversity values within the caves of the PNKBNP and enable adequate protection further, more comprehensive surveys must be undertaken across several caves in the two main karst drainage systems identified (Figure 2). This will likely lead to the addition of ‘Biodiversity Values’ for the recognised World Heritage values of the park.
In addition to further research programmes in the caves, the currently known cave biota must be adequately protected and managed. This requires some immediate actions in the current tourist caves to stop the destruction of key cave fauna habitats, including:
- Immediately make defined pathways in the tourist caves marked by posts and rope to stop the cave floors being destroyed by people walking on them. This destroys habitat for many insects and spiders that live in the cave. Ultimately raised walkways should be installed in all tourist areas to minimise impact to cave floors.
- Clean the compacted mud from the cave floor to allow habitat for cave insects and spiders to recover.
- Clean up the rubbish left throughout the cave. Rubbish attracts rats into the cave that will eat cave insects and spiders.
- Remove the rubbish bins which attract rats (see above), and also attracts surface insects which are affecting the diversity and abundance of cave insects and spiders.
- Stop people eating and drinking inside the caves as food scraps dropped also encourage rats to live in the caves.
- Reduce noise in the caves as this is disturbing the bat and swiftlet populations which support diverse insect communities in the caves. If the noise continues, the bats and swiftlets may leave the cave permanently, destroying the cave guano insect ecosystem.
- Enforce the ‘No Smoking’ ban inside the caves as this is also disturbing the bat and swiftlet populations, and also increases rubbish dropped on floors as butts and empty packets.
- Change the lighting in the caves as it is causing the growth of lampenflora (plants that grow under artificial light in caves). The lampenflora is providing an artificial food source for surface species not usually found in caves, and thus affecting the diversity and abundance of cave insects and spiders.
- Additional, more comprehensive biodiversity surveys are required to able to fully appreciate the diversity of cave fauna in the PNKBNP. This will provide a far greater understanding of biodiversity and the unique species present here.
- Prior to the development of additional caves for tourism, they must be comprehensively assessed for cave biodiversity to provide baseline data to enable annual monitoring of impacts to fauna. The baseline survey will also identify any key habitats to be preserved within each cave, and any important species that may require special protection.
The implementation of these simple and generally low cost options will dramatically improve the habitat for cave fauna and allow a more natural community to re-establish within the highly impacted areas of Phong Nha Cave system (Bi Ki Grotto, Fairy Grotto, and Royal Grotto areas).
The caves of the Phong Nha – Ke Bang National Park are highly likely contain a very important and diverse community of cave fauna. This preliminary survey provides only a small insight into the biological treasures to be discovered by future research in this rich and unique World Heritage Site.
Acknowledgements
The authors would like to acknowledge the Peoples Republic of Vietnam for the opportunity to visit their beautiful country and the very warm welcome received. This preliminary survey would not have been possible without the co-operation of the Phong Nha – Ke Bang National Park, the Phong Nha – Ke Bang Tourism Centre and the Scientific Research and Wildlife Rescue Centre. The authors wish to thank the Phong Nha – Ke Bang National Park Region Project, supported by the German Ministry of Cooperation and Development (BM) for supporting this important project to begin the understanding of cave biodiversity in PNKBNP. Special thanks to all the GTZ - staff in Dong Hoi for their tireless efforts to make this survey possible and make us all feel extremely welcome.

Figure 14 The authors (left to right), Renee Mouritz, Pham Dinh Sac, and Timothy Moulds in Bi Ki Grotto, Phong Nha Cave. (Photo Dang Ngoc Kien).
References
Barr, T. C. J. (1968). Cave ecology and the evolution of troglobites. Evolutionary Biology 2. Dobzhansky, T., Hect, M. and Steere, W. New York, Appleton-Century-Crofts. 2: 35-102.
Blyth, J., Jasinska, E., Mutter, L., English, V. and Tholen, P. (2002). Threatened wildlife of the Yanchep Caves. Landscope 17: 34-40.
Boutin, C. (2004). Organisms: classification. Encyclopedia of caves and karst science. Gunn, J. London, Fitzroy Dearborn: 548-550.
Christiansen, K. A. (1962). Proposition pour la classification des animaux cavernicoles. Spelunca Mem. 2: 76-78.
Clark, B. (2009a). A conceptual development plan for show caves in the Phong Nha – Ke Bang National Park in the Quang Binh Province, Vietnam. Unpublished report to GTZ, April 2009, pp. 18.
Clark, B. (2009b). Report to the provincial project management unit in regard to the nature conservation and sustainable management of natural resources in the Phong Nha – Ke Bang National Park Region. Unpublished report to Provincial Management Unit, Quang Binh Province Vietnam, October 2009, pp. 30.
Culver, D. C., Kane, T. C. and Fong, D. W. (1995). Adaptation and natural selection in caves: the evolution of Gammarus minus. Cambridge MA, Harvard University Press.
Decu, V. (1986). Some considerations on the bat guano synusia. Travaux de l'Institut de Spéologie "Emile Racovitza" 25, 41-51.
Ferreira, R. L. and Martins, R. P. (1998). Diversity and distributions of spiders associated with bat guano piles in Mirrinho Cave (Bahia State, Brazil). Diversity and Distributions 4: 235-241.
Ferreira, R. L. and Martins, R. P. (1999). Trophic structure and natural history of bat guano invertebrate communities, with special reference to Brazilian caves. Tropical Zoology 12: 231-252.
Gnaspini, P. (1992). Bat guano ecosystems. A new classification and some considerations, with special references to Neotropical data. Mémoires de Biospéologie 19, 135-138.
Gnaspini, P. and Trajano, E. (2000). Guano communities in tropical caves. In 'Ecosystems of the world. Subterranean ecosystems'. (Eds H. Wilkens, D. C. Culver and W. F. Humphreys). 251-268. (Elsevier: Amsterdam)
Harris, J. A. (1970). Bat guano cave environment. Science 169, 1342-1343.
Hawes, R. S. (1939). The flood factor in the ecology of caves. Journal of Animal Ecology 8: 1-5.
Hoch, H. (1988). A new cavernicolous planthopper species (Homoptera: Fulgoroidea: Cixiidae) from Horst, R. (1972). Bats as primary producers in an ecosystem. Bulletin of the National Speleological Society 34: 49-54.
Hoch, H. and Howarth, F. G. (1999). Multiple cave invasions by species of the planthopper genus Oliarus in Hawaii (Homoptera: Fulgoroidea: Cixiidae). Zoological Journal of the Linnean Society 127, 453-475.
Horst, R. (1972). Bats as primary producers in an ecosystem. Bulletin of the National Speleological Society 34: 49-54.
Howarth, F. G. (1973). The cavernicolous fauna of Hawaiian lava tubes, 1. Introduction. Pacific Insects 15: 139-151.
Howarth, F. G. and Stone, F. D. (1990). Elevated carbon dioxide levels in Bayliss Cave, Australia: Implications for the evolution of obligate cave species. Pacific Science 44: 207-218.
Howarth, F. G. (2003). Cave insects. Encyclopedia of Insects. Resh, V. H. and Carde, R. T. San Diego, Elsevier. 1: 1-1266.
Humphreys, W. F. (1991). Experimental re-establishment of pulse-driven populations in a terrestrial troglobite community. Journal of Animal Ecology 60: 609-623.
Humphreys, W. F. (2000). Background and glossary. Ecosystems of the world. Subterranean ecosystems. Wilkens, H., Culver, D. C. and Humphreys, W. F. Amsterdam, Elsevier. 30: 3-14.
Humphreys, W. F. and Eberhard, S. (2001). Subterranean fauna of Christmas Island, Indian Ocean. Helictite 37: 59-73.
Koon, L. C. and Cranbrook, E. (2002). Swiftlets of Borneo. Builders of edible nests, Natural History Publications (Borneo).
Lamprecht, G. and Weber, F. (1992). Spontaneous locomotion behaviour in cavernicolous animals: the regression of the endogenous circadian system. The natural history of biospeleology. Camacho, A. I. Madrid, Spain, National Museum of Natural Sciences: 225-262.
Langecker, T. G. (2000). The effects of continuous darkness on cave ecology and cavernicolous evolution. Subterranean ecosystems. Wilkens, H., Culver, D. C. and Humphreys, W. F. Amsterdam, Elsevier. 30: 135-157.
Medway, L. (1962). The swiftlets (Collocalia) of Niah Cave, Sarawak. Part 2 Ecology and the regulation of breeding. Ibis 104: 228-245
Moulds, T. A. (2004). Review of Australian cave guano ecosystems with a checklist of guano invertebrates. Proceedings of the Linnean Society of New South Wales 125, 1-42.
Moulds, T. A. (2006). The diversity, seasonality, and ecology of cavernicolous guano dependant arthropod ecosystems in southern Australia. Unpublished PhD thesis, The University of Adelaide, pp 250.
Peck, S. B. (1976). The effect of cave entrances on the distribution of cave inhabiting terrestrial arthropods. International Journal of Speleology 8: 309-321.
Poulson, T. L. and White, W. B. (1969). The cave environment. Science 165: 971-981.
Poulson, T. L. (1972). Bat guano ecosystems. Bulletin of the National Speleological Society 34: 55-59.
Poulson, T. L. and Lavoie, K. H. (2000). The trophic basis of subsurface ecosystems. In 'Ecosystems of the world. Subterranean ecosystems'. (Eds H. Wilkens, D. C. Culver and W. F. Humphreys). 231-249. (Elsevier: Amsterdam).
Poulson, T. L. (2005). Food sources. Encyclopedia of caves. Culver, D. C. and White, W. B. San Diego, California, Elsevier. 1: 255-263.
Richards, A. M. (1971). An ecological study of the cavernicolous fauna of the Nullarbor Plain Southern Australia. Journal of the Zoological Society of London 164, 1-60.
Racovitza, E. G. (1907). Essai sur les problemes biospeologiques. Arch. zool. exp. et gen. 36: 371-488.
Schiner, J. R. (1854). Fauna der Adelsberger, Lueger- und Magdalener-grotte. Die grotten und hohlen von Adelsberg, Lueg, Planina und Lass. Schmidl, A. Wien, Braunmuller: 231-272.
Trajano, E. (1996). Movements of cave bats in southeastern Brazil, with emphasis on the population ecology of the common vampire bat, Desmodus rotundus (Chiroptera). Biotropica 28: 121-129.
Volschenk, E. S. and Prendini, L. (2008). Aops oncodactylus, gen. et sp. nov., the first troglobitic urodacid (Urodacidae: Scorpiones), with a re-assessment of cavernicolous, troglobitic scoprions. Invertebrate Systematics 22: 235-257.